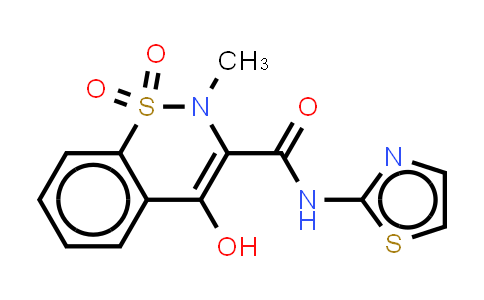
| Chemical Name |
Sudoxicam |
| CAS Number |
34042-85-8 |
| MDL Number |
MFCD00866053 |
| Molecular Formula |
C13H11N3O4S2 |
| Molecular Weight |
337.37 |
Introduction of 34042-85-8 :
Sudoxicam is a reversible and orally active COX antagonist and a non-steroidal anti-inflammatory drug (NSAID) from the enol-carboxamide class. Sudoxicam has potent anti-inflammatory, anti-edema and antipyretic activity[1][2][3]. IC50 & Target: COX[3] In Vitro: Sudoxicam demonstrates NADPH-dependent covalent binding to human liver microsomes. With addition of glutathione (GSH) in microsomal incubations, about half of the covalent incorporation of Sudoxicam is blocked by addition of GSH[1].
Metabolite identification studies on [14C]-Sudoxicam in NADPH-supplemented human liver microsomes indicated that the primary route of metabolism involved a P450-mediated thiazole ring scission to the corresponding acylthiourea metabolite (S3), a well-established pro-toxin[1].
In vitro, Sudoxicam underwent the oxidative thiazole-open biotransformation, resulting in the formation of an acylthiourea and the subsequent hydroxylated metabolite[3]. In Vivo: Sudoxicam (1-10 mg/kg; oral administration; daily; for 7 days; rats) treatment effective reduces plasma inflammation units, reduces the swelling of inflamed hind-paws and restores toward normal the daily gain in body weight[2].
In the intact rat, Sudoxicam significantly inhibited edema formation at doses as low as 0.1 mg/kg, p.o[2].
Sudoxicam inhibits the erythema caused by ultraviolet irradiation in the guinea pig. Sudoxicam (3.3 mg/kg, i.p.) is capable of counteracting the pyrexia induced by the intraperitoneal injection of typhoid/paratyphoid vaccine in rats, maintaining body temperature about that of uninjected control rats[2].
The plasma half-life of Sudoxicam ranged between 8 hours (monkey), 13 hours (rat), and 60 hours (dog)[2].
| Purity |
NLT 98% |
| Storage |
at 20ºC 2 years |
*The above information is for reference only.